[insight] The Significance of Precision Weighing in Analytical Chemistry
{:en}[vc_row][vc_column][vc_column_text] Precision weighing is a fundamental step in analytical chemistry, as the accuracy of sample mass directly influences calculations and analytical outcomes. Even minor deviations during weighing can lead to significant errors in data interpretation and final results. Therefore, reliable weighing instruments and proper handling procedures are essential to ensure data integrity. In advanced analytical applications, particularly those involving micro-scale samples, semimicro balances play a critical role. These balances provide exceptional accuracy at the microgram level, enabling analysts to measure very small sample quantities with confidence. Their high sensitivity and stability make them indispensable in research and quality control laboratories. By utilizing semimicro
[guide] Tips for Setting Accurate Temperature Ranges for Lab Applications
{:en}[vc_row][vc_column][vc_column_text] Maintaining precise temperature control is essential in laboratory environments, especially for sensitive experiments and sample preservation. The first step is to carefully program the desired temperature range according to the specific requirements of your application. Different research processes may require variations, so accurate input ensures optimal performance. Before starting, it is important to pre-check calibration settings. Regular calibration guarantees that instruments deliver reliable and reproducible results. Skipping this step may cause deviations that compromise both data accuracy and experiment integrity. Ensuring equipment is properly calibrated helps maintain confidence in laboratory outcomes. Lastly, continuous monitoring of real-time temperature readings is key to consistency.
[remark] Advancements in Heating and Cooling Block Technology
{:en}[vc_row][vc_column][vc_column_text] Heating and Cooling Block technology has evolved to meet the growing demands of complex molecular biology and biochemical research. These systems are engineered for rapid and precise temperature adjustments, ensuring uniform thermal treatment across all samples. This leads to improved data reproducibility and more efficient workflows. A key innovation is the uniform temperature distribution across the block, even during rapid shifts in temperature. This fast response makes the system ideal for sensitive applications like enzymatic reactions, PCR preparation, or RNA/DNA extraction, where thermal consistency is critical. The modular design of modern Heating and Cooling Blocks allows researchers to process multiple samples under
[insight] Precision Temperature Control for Enzymatic Reactions and Protein Studies
{:en}[vc_row][vc_column][vc_column_text] In biochemical and molecular biology research, precise temperature control is crucial, particularly in temperature-sensitive applications like enzymatic reactions and protein crystallization. Even slight temperature deviations can alter reaction kinetics or compromise the structural integrity of delicate proteins, leading to inconsistent or unreliable results. Heating and Cooling Blocks are designed to deliver uniform thermal regulation across all samples. With digital controls and rapid heat exchange capabilities, these systems ensure that target temperatures are reached and maintained with exceptional accuracy. This stability is essential for achieving reproducibility in high-sensitivity experiments. By using reliable thermal equipment, laboratories can maintain strict environmental parameters, protecting sample integrity
[guide] Best Practices for Using an Automatic Media Preparator
{:en}[vc_row][vc_column][vc_column_text] Using an automatic media preparator correctly is key to ensuring consistent, sterile, and high-quality culture media. The process begins with selecting the appropriate type of culture medium based on your microbiological testing needs. After preparing the solution, it is loaded into the media preparator for automated processing. Once the media is inside the machine, users can set the desired temperature and mixing parameters. These settings are critical to ensure homogeneity and to prevent uneven heating or sedimentation. The sterilization step follows, during which the machine heats the solution to eliminate all microbial contaminants, ensuring the medium is ready for reliable laboratory
[remark] Automatic System for Dispensing Culture Media
{:en}[vc_row][vc_column][vc_column_text] In microbiological laboratories, consistent quality in culture media preparation is essential. Our Media Preparator offers a fully automated system for both preparing and dispensing culture media, significantly reducing manual handling and the risk of contamination. This automation ensures each batch is processed under uniform conditions, improving overall reliability. Equipped with adjustable temperature and continuous stirring controls, the system maintains precise thermal stability and media homogeneity. These features are critical in preserving the integrity of the culture medium, allowing for accurate microbial growth and consistent results across different tests and applications. Designed for high-throughput environments, the Media Preparator supports time efficiency and standardization
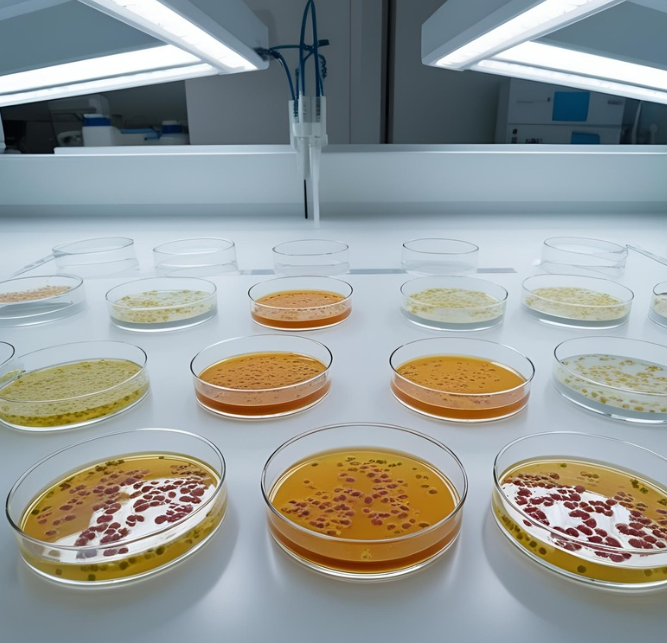
[insight] The Importance of Sterile Culture Media in Microbiological Studies
{:en}[vc_row][vc_column][vc_column_text] Sterile culture media are fundamental to microbiological studies, serving as the primary environment for microbial growth and analysis. Any contamination in the media can introduce unwanted variables, leading to inaccurate results and potentially misleading conclusions. Maintaining sterility is therefore critical for ensuring the reliability and reproducibility of research data. In high-throughput environments such as pharmaceutical and clinical laboratories, manual media preparation can be both time-consuming and error-prone. Automated media preparators offer a streamlined solution by ensuring precise temperature control, homogeneity, and consistent sterilization—factors that are essential for high-quality microbiological testing. By adopting automated systems, laboratories can improve efficiency, reduce human error, and
[guide] Conducting Compliance Testing for Tablet Disintegration
{:en}[vc_row][vc_column][vc_column_text] Compliance testing for tablet disintegration is an essential step in pharmaceutical quality assurance. The process begins by preparing the disintegration media in accordance with USP guidelines, ensuring standardized and repeatable conditions for every test. Using the correct temperature and medium composition is vital to obtain accurate and valid results. Once the medium is ready, the tablets are carefully placed into the disintegration baskets, and the test is initiated using a calibrated disintegration tester. The equipment simulates the conditions in the human gastrointestinal tract, allowing manufacturers to evaluate whether tablets will break down within the expected timeframe under physiological conditions. Throughout the process,
[remark] Accuracy and Reliability in Tablet Disintegration Testing Equipment
{:en}[vc_row][vc_column][vc_column_text] Accuracy and reliability are critical in tablet disintegration testing to ensure the quality and consistency of pharmaceutical products. Our Tablet Disintegration Tester is engineered to meet USP (United States Pharmacopeia) standards, delivering dependable and reproducible results for every test. Equipped with an intuitive interface and user-friendly controls, this device enables laboratory technicians to perform tests with high efficiency. Its robust construction and durable materials make it a long-term, low-maintenance solution for pharmaceutical laboratories demanding consistent performance. Reliable disintegration testing supports not only regulatory compliance but also patient safety. By using accurate and stable testing equipment, manufacturers can ensure each tablet disintegrates as
[insight] Understanding the Disintegration Process in Drug Formulation
{:en}[vc_row][vc_column][vc_column_text] In the pharmaceutical field, tablet disintegration is a key parameter in drug formulation. Disintegration refers to a tablet’s ability to break down into smaller particles once ingested, prior to dissolving in bodily fluids. This process ensures the active ingredient is released at the right time and in a form that can be readily absorbed. The speed and effectiveness of disintegration significantly impact a drug's bioavailability—how much and how quickly the active compound reaches systemic circulation. If a tablet fails to disintegrate properly, absorption may be delayed or incomplete, reducing therapeutic efficacy. This is why disintegration testing is a crucial part of